Thermal Phenomena in Induction Heating: Conductivity, Heat Capacity, and Why ‘Uniform’ Is Hard
14 min
- Introduction to Thermal Dynamics in Induction Heating
- The Physics of Heat Transfer: Conduction, Convection, and Radiation
- Thermal Conductivity (k): The Speed of Heat
- The Relationship Between Thermal and Electrical Conductivity
- Heat Capacity (C) and Specific Heat (c): The Energy Storage
- Summary of Practical Implications for Induction Engineering
- Final Engineering Checklist for Thermal Review
- FAQ
Introduction to Thermal Dynamics in Induction Heating
In the precision-driven field of induction heating (IH), we often focus our primary engineering attention on the electromagnetic side of the equation. We calculate skin depth, frequency, and coil coupling with meticulous care, ensuring that the induced eddy currents are generated precisely where they are needed. However, once those currents are induced and the energy is successfully absorbed within the workpiece, the laws of electromagnetics hand the baton to the laws of thermodynamics. It is here, in the thermal regime, where the success of a process is truly determined. Understanding the thermal phenomena that govern heat flow and storage is not merely a theoretical exercise; it is a practical necessity for preventing distortion, ensuring metallurgical integrity, and achieving energy efficiency in modern manufacturing.
The challenge of induction heating is that we are typically dealing with moving targets. Unlike a static furnace environment where materials might eventually reach a steady-state equilibrium after hours of soaking, induction heating is often rapid, intense, and localized. This creates significant temperature gradients between the surface and the core, or between the target heating zone and the rest of the component. In this high-intensity environment, two fundamental material properties—thermal conductivity and heat capacity—dictate how the energy we've pumped into the material will redistribute itself across the workpiece. As we will explore in detail, these properties are rarely constant; they shift and change as the material heats up, creating a complex, nonlinear puzzle that the engineer must solve to achieve repeatable, high-quality results. This blog post explores these critical phenomena, grounded in the empirical data and physical laws that define modern induction heating practice.
The Physics of Heat Transfer: Conduction, Convection, and Radiation
In any induction heating application, all three modes of heat transfer are present and interacting. While conduction occurs within the solid mass of the workpiece, convection and radiation act at the surface, often representing the primary sources of energy loss to the surroundings. For a successful process, the engineer must not only manage the heat we want inside the part (internal conduction) but also mitigate and account for the heat we lose to the environment (surface convection and radiation). Each of these modes follows distinct physical laws that must be integrated into the process design.
Internal Conduction and Fourier's Law
Thermal conduction is the mechanism by which heat is transferred from high-temperature regions of the workpiece toward low-temperature regions. This internal flow is described by Fourier's law, which states that the heat flux by conduction (qcond) is proportional to the thermal conductivity (k) and the temperature gradient (grad T). In essence, heat flows from hot to cold at a rate determined by the material's conductive "speed." Mathematically, this is expressed as:
For an engineer, this equation reveals a fundamental truth: the rate of heat transfer increases with both the "steepness" of the temperature difference and the material's ability to conduct. During surface hardening, for instance, we intentionally create a massive temperature gradient between the hot surface (which may be at 900°C) and the cold core (at room temperature). If the material's thermal conductivity is high, heat will race toward the core with incredible intensity. Conversely, the rate of transfer is inversely proportional to the distance—the further the heat has to travel, the slower the core will respond. This relationship is the primary reason why controlling the time of the heating cycle is as critical as controlling the power level; the "soak time" must be precisely balanced against the conductive flow.
Surface Losses: Convection and Radiation
While we want to control the heat inside the metal, we are also fighting to keep it from escaping. Convection heat transfer is carried out by the fluid, gas, or air surrounding the workpiece. According to Newton's law, the heat transfer rate is directly proportional to the temperature difference between the surface of the workpiece and the ambient area. In many induction heating applications, such as the continuous heating of strips, wires, or rotating shafts, we cannot assume "free" or natural convection. Forced convection—caused by the movement of the part itself or the high-velocity flow of cooling air—can increase heat losses dramatically. In some strip coating applications where the metal travels at speeds up to 5 m/s, the heat losses attributed to forced convection are often 5 to 10 times higher than those of a stationary part.
Thermal radiation is even more aggressive as temperatures climb. Governed by the Stefan–Boltzmann law, radiation losses are proportional to the fourth power of the absolute temperature. This means that at the high temperatures required for hot working, forging, or the heating of titanium and tungsten (e.g., above 1200°C), radiation becomes the dominant source of heat loss. The efficiency of the process is then heavily tied to the material's emissivity—a coefficient that accounts for the metal's surface condition. Polished metals radiate significantly less heat than those with oxidized or non-polished surfaces. For example, a carbon steel slab at 1250°C with an emissivity of 0.8 can lose heat at a density of 244 kW/m², a staggering amount of energy that must be compensated for by the induction power supply just to maintain the target temperature.
Thermal Conductivity (k): The Speed of Heat
Thermal conductivity (k) is the property that designates the rate at which heat travels across a thermally conductive workpiece. It is effectively the material's "thermal bandwidth." A material with a high k value conducts heat much faster than one with a low k. In the design of induction heating inductors, for example, we require materials for refractories or liners that have a very low k to minimize surface heat losses and maximize thermal efficiency. However, when we look at the workpiece being heated, the value of k can be either a friend or a foe, depending entirely on the metallurgical goal of the operation.
When High Conductivity is Beneficial: Through-Heating
In through-heating applications, the engineering goal is to achieve a uniform temperature throughout the entire volume of the material. This is common in forging, rolling, or annealing. Here, a high value of k is an advantage because it facilitates the rapid migration of heat from the surface (where it is generated by induction skin effects) into the core. This helps prevent the surface from overheating while waiting for the center to reach the target temperature, ultimately leading to a more uniform and predictable thermal profile. Without sufficient conductivity, the surface could melt before the core even reaches its transformation temperature, especially in large-diameter billets.
When High Conductivity is Problematic: Selective Heating
The script flips when we move to selective heating applications, such as gear surface hardening or the case hardening of shafts. In these scenarios, we want the heat to stay exactly where it was generated—typically in a thin "case" on the surface, leaving the core tough and unhardened. A high value of k becomes a significant disadvantage because it promotes heat transfer and works to equalize the temperature distribution within the workpiece. This "heat spreading" carries energy away from the target hardening zone and into adjacent areas that were intended to remain cool.
This unintended heat transfer has several practical engineering consequences that can compromise the final product:
- Reduced Energy Efficiency: Heating mass that does not need to be hardened is a direct waste of power, increasing the cost per part and the carbon footprint of the process.
- Metallurgical Degradation: The temperature rise in adjacent areas can negatively affect the microstructural characteristics of the steel, potentially tempering or softening areas that require specific mechanical properties.
- Residual Stress and Distortion: Heating a larger volume of material than necessary increases the magnitude of thermal expansion and subsequent contraction. This is a primary driver of excessive distortion and can lead to unfavorable residual stress profiles, requiring expensive post-process straightening or grinding.
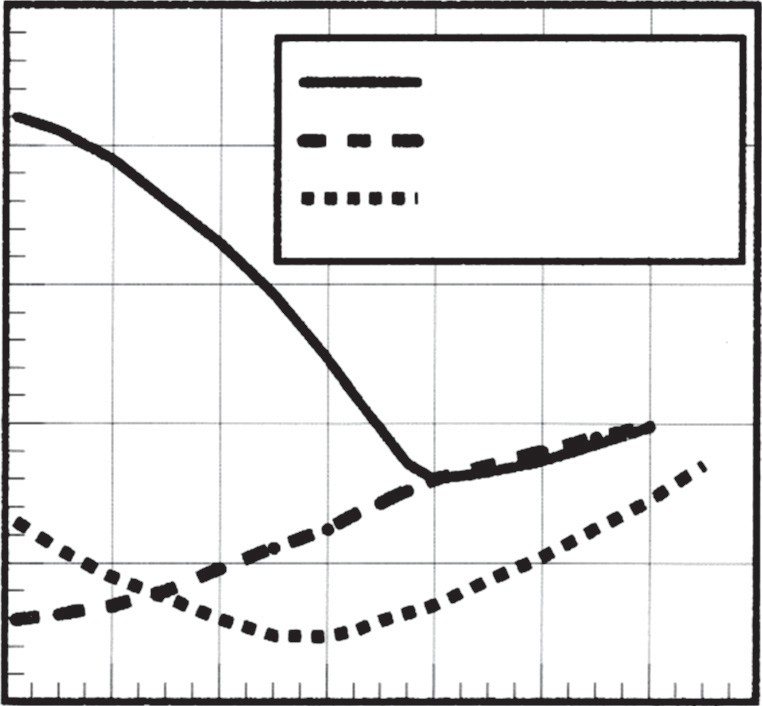
The Relationship Between Thermal and Electrical Conductivity
There is a deep physics-based link between how a metal conducts electricity and how it conducts heat. For the majority of pure metals and metallic materials, this relationship is governed by the Wiedemann–Franz law. This law governs the relationship between thermal conductivity (κ) and electrical conductivity (σ) for metallic materials, which is also a function of the temperature (T). Generally, if a material is an excellent electrical conductor (meaning it has low resistivity), it is also likely to be an excellent thermal conductor. Copper and aluminum are classic examples of materials that follow this trend, being excellent at conducting both current and heat.
However, engineers must be wary of simple generalizations. Some alloys, notably cast irons, can be significant exceptions to the Wiedemann–Franz law. Furthermore, the presence of alloying and residual elements can have a measurable impact on thermal conductivity. As shown in the data for 7000-series aluminum alloys, the presence of alloying elements typically lowers both the electrical and thermal conductivity compared to the pure metal base. For example, alloy 7075-T6 has an electrical resistivity 93% greater than pure aluminum, and its thermal conductivity is correspondingly reduced. Even the temper treatment (e.g., O-temper vs T6) can shift these properties significantly.
| Material Condition | Elect. Resistivity [μΩ·m] | Thermal Conductivity [W/(m·°C)] | Specific Heat [J/(kg·°C)] | |
|---|---|---|---|---|
| 1 | Pure Aluminum (Commercial) | 0.027 | 211 | 933 |
| 2 | Alloy 7005 (O-Temper) | 0.04 | 166 | 855 |
| 3 | Alloy 7075 (T6-Temper) | 0.052 | 130 | 960 |
Heat Capacity (C) and Specific Heat (c): The Energy Storage
While thermal conductivity tells us how fast heat moves, heat capacity tells us how much energy we need to put in to get a result. The heat capacity (C) is a measure of the amount of energy that would have to be absorbed by the workpiece to achieve a unit of required temperature change. It is measured in units of J/(mol °C) and is defined mathematically by the relationship:
In most practical engineering calculations, we use specific heat (c), which represents the heat capacity per unit mass. This is the amount of energy required to raise a unit mass of the material by one unit of temperature (e.g., 1 kg by 1°C). The units are typically J/(kg °C) or Btu/(lb °F). This property is what defines the "thermal mass" of the part and determines the total energy budget for the heating cycle.
Power Implications for the Designer
The engineering significance of specific heat is direct and unavoidable: a higher value of c corresponds directly to the greater required power to heat a unit mass to a unit temperature. Materials with high specific heat are effectively "thermal sponges"; they soak up vast amounts of energy with relatively small increases in temperature. When designing an induction heating system, the power supply must be sized not just for the electromagnetic coupling efficiency, but for the fundamental energy "appetite" of the material. If the power density is too low for a material with high c, the part may never reach the target temperature because the energy is being radiated or convected away as fast as it is being absorbed.
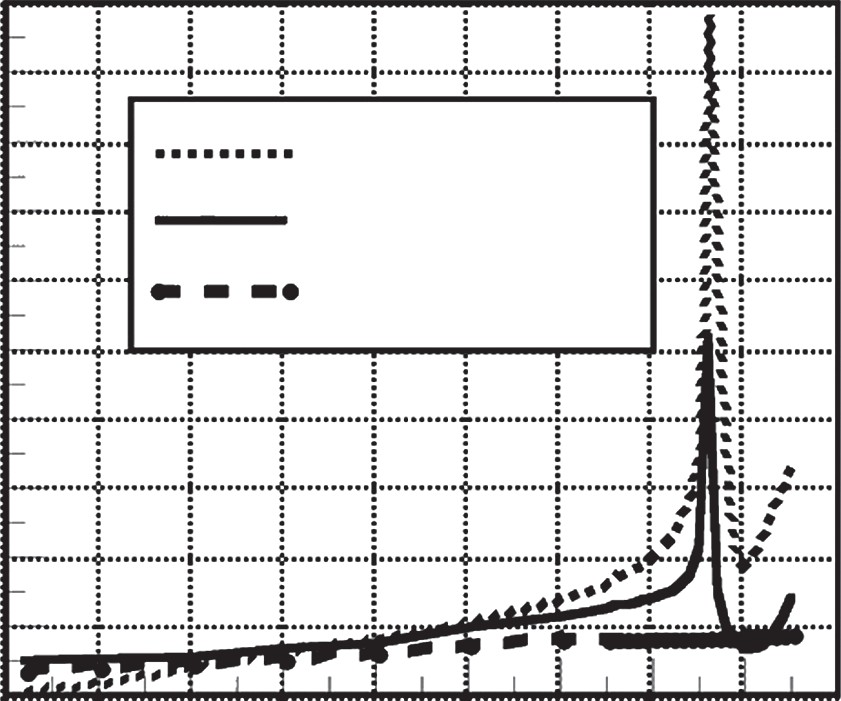
Temperature Dependence of Specific Heat
As illustrated in Figure 3, specific heat is a highly temperature-dependent property that does not follow a linear path for all materials. For pure metals such as copper and aluminum, the increase in specific heat with rising temperature is relatively gradual and predictable. However, for plain carbon steels (such as SAE 1010 and 1042) and stainless steels (like 304), the specific heat exhibits dramatic, nonlinear spikes at specific temperature ranges. According to the data in Figure 3.49(b) from the reference material, these materials require significantly more energy to achieve a unit of temperature change as they reach critical thermal points.
This nonlinear behavior has immediate practical implications: an induction heating cycle cannot be treated as a simple constant-power ramp. The power density that is sufficient to raise the temperature in a lower range may be completely inadequate to maintain the heating rate once the material enters these high-energy-absorption zones. Engineers must account for these "thermal hurdles" when designing the power profile of a cycle. If the power supply does not have sufficient headroom or if the system is not programmed to adapt its power output, the heating rate will stall or slow down significantly at these critical temperatures, leading to inconsistent cycle times and potential quality failures.
Summary of Practical Implications for Induction Engineering
The interaction between thermal conductivity and specific heat determines the final thermal profile and metallurgical quality of the workpiece. When these properties vary—due to temperature, alloying, or residual elements—the entire induction process shifts. Ignoring these nonlinearities leads to inconsistent results, from variations in case depth to unexpected metallurgical failures and excessive part distortion.
Practical takeaways for the process engineer include:
- Strategic Process Selection: High thermal conductivity is beneficial for through-heating but detrimental for selective surface hardening. For selective hardening, you must use high power densities and extremely short cycle times to "beat" the thermal conductivity and keep the heat localized.
- Material Sensitivity: Don't rely on generic material data. Residual elements and specific temper treatments can shift thermal conductivity by 30-40% compared to pure metal baselines. Always verify property data for the exact grade and condition of your workpiece.
- Dynamic Power Cycles: Specific heat spikes at critical temperatures mean that constant power heating is rarely ideal. Adaptive power cycles that account for these transitions are necessary for both precision and energy efficiency.
- Controlling Distortion: Excessive heating of adjacent zones due to high k is a primary driver of part distortion and unfavorable residual stress. Manage this by using localized coils and rapid, high-intensity heating cycles.
- Accounting for Surface Losses: Radiation and convection losses increase dramatically with temperature and surface condition. The power required to maintain a temperature in a forging billet is often a significant fraction of the initial heating power.
Understanding these thermal foundations ensures that induction heating remains a precision tool rather than a source of manufacturing variability. By accounting for the way heat moves and is stored within the material, engineers can design more efficient, repeatable, and high-quality thermal processes that meet the demanding requirements of modern high-performance manufacturing.
Final Engineering Checklist for Thermal Review
- Analyze the thermal conductivity profile of the specific alloy across the full temperature range, noting any nonlinear drops or spikes.
- Calculate the total energy requirement based on mass and temperature-dependent specific heat, accounting for transformation energy hurdles.
- Assess convection and radiation losses at the target temperature to ensure the power supply has sufficient headroom for maintenance.
- Review the impact of alloying elements and heat treatment on both electrical resistivity and thermal properties compared to pure metal baselines.
- Evaluate the potential for heat spreading in selective hardening applications and adjust cycle times to minimize unintended heat migration into adjacent zones.
FAQ
Q: Why is thermal conductivity both helpful and problematic in induction heating?
It depends on your goal. For through-heating applications like forging or annealing, high thermal conductivity is beneficial because it helps heat migrate quickly from the surface to the core, creating uniform temperature distribution. However, for selective surface hardening (like gear hardening), high conductivity becomes a problem—it spreads heat away from your target zone into areas you want to keep cool, wasting energy and potentially causing distortion or unwanted metallurgical changes.
Q: Why can't I use constant power throughout my heating cycle?
Because specific heat isn't constant—it changes dramatically with temperature, especially in carbon and stainless steels. These materials hit "thermal hurdles" at critical temperatures where they suddenly require much more energy per degree of temperature rise. If you don't increase power at these points, your heating rate will stall. You need adaptive power cycles that account for these nonlinear energy absorption zones to maintain consistent heating rates and quality.
Q: How do surface losses affect my power requirements at high temperatures?
Surface losses become massive at elevated temperatures, especially radiation losses which scale with the fourth power of absolute temperature. At forging temperatures (above 1200°C), radiation can dominate your energy budget—a carbon steel slab at 1250°C can lose 244 kW/m² just from radiation alone. This means a significant portion of your power supply capacity isn't heating the part, it's just compensating for surface losses. Surface condition matters too: oxidized surfaces radiate far more heat than polished ones.
Keep Learning
Heating Slabs, Blooms, and Rectangular Bars by Induction: Edge Control Is Everything
Key Takeaways Edge control is critical: Slabs, blooms, and rectangular bars exhibit complex electromagnetic edge and end effects that directly impact heating uniformity and product quality. Frequency selection matters: The optimal skin-depth-to-thickness ratio ($d/\delta$) of approximately 3–3.5 for nonmagnetic slabs balances penetration depth, heating uniformity, and coil electrical efficiency. Coil overhang optimization: A normalized coil overhang of about $\sigma/d \approx 0.7$ creates balanced pow......
Induction Heating for Semisolid Processing: Billet and Slug Recipes That Work
Key Takeaways Semisolid Temperature Window: Aluminum alloys require heating to 575°C–595°C with ±3°C to ±4°C uniformity for consistent 50% liquid fraction and high-quality castings. Billet Geometry Matters: Maintain slug length-to-diameter ratios between 1:2 and 1:3 to prevent segregation and ensure uniform heating throughout the workpiece. Multistage Heating Is Essential: Combine rapid power-coil heating with soaking in holding coils to homogenize temperature and microstructure before casting. Freque......
Billet Heating and End Heating by Induction: Uniformity, Throughput, and Coil Strategy
Key Takeaways Temperature Targets: Steel billets require heating to 1050°C–1260°C with surface-to-core uniformity within ±20°C to ±30°C for quality forging outcomes. Frequency Matters: Billet diameter dictates frequency selection—higher frequencies (>30 kHz) for small billets, lower frequencies (<500 Hz) for large billets—to optimize heating depth and uniformity. Profiled Coils Outperform: Variable-pitch, graded coils deliver superior temperature uniformity and shorter coil lengths compared to convent......
In-Line Induction Heating of Bars and Rods: Continuous Throughput Without Temperature Surprises
Key Takeaways Electromagnetic Skin Effect: During initial heating, eddy currents concentrate in a thin surface layer, creating steep surface-to-core temperature gradients that must be managed carefully. Arcing Prevention: Longitudinal voltage drops along bars can cause arcing with support rolls. Alternating coil winding directions or tailored electrical connections are key mitigation strategies. Copper Shield Plates: Unslotted copper end plates act as Faraday rings to reduce electromagnetic cross-talk......
Fundamentals and Design Approaches for Induction Mass Heating
Key Takeaways Frequency is critical: The workpiece diameter-to-penetration depth ratio must exceed four to prevent eddy current cancellation and ensure efficient, uniform heating through the cross section. Material-specific challenges: Stainless steels and superalloys demand tighter control due to high electrical resistivity and low thermal conductivity, making cross-sectional uniformity harder to achieve. Four heating modes: Engineers select from static, progressive multistage, continuous, and oscill......
Advanced Induction Joining and Assembly: From Adhesive Bonding to Motor Rotor Integration
Key Takeaways Induction Bonding: Uses heat transferred from an induction-heated metallic part to cure adhesives, enabling strong joints between dissimilar materials like plastics, ceramics, and metals. Cap Sealing: A non-contact induction process that heats an aluminum foil liner inside bottle caps to create hermetic, tamper-evident seals at high line speeds. Shrink Fitting: Exploits thermal expansion to create high-strength interference fits without fasteners or fillers, and is often reversible for m......