The Silent Destroyer: Why Corrosion is More Than Just Rust
8 min
- The High Cost of Decay
- The Science: Nature’s Invisible Battery
- The Shield: Passivation
- The Rogues’ Gallery: How Corrosion Attacks
- The Achilles’ Heel: Welding
- Winning the War: Prevention and Control
- Conclusion
- FAQ
Picture a rusty patch on an old car bumper or a seized bolt in the garden shed. Easy to dismiss as a cosmetic nuisance. But step inside a chemical refinery, an offshore oil platform, or a nuclear power plant, and corrosion transforms from eyesore to existential threat. It is a relentless, multi-billion-dollar adversary that engineers wage a daily war against — one they can never fully win, only manage.
To an engineer, corrosion is the destructive deterioration of a material as it reacts with its environment. Put more poetically: it is nature’s slow, patient effort to reclaim what humans have refined. Every pipe we lay, every vessel we weld, every bridge we build is, thermodynamically speaking, already on its way back to ore. Understanding that journey — and how to slow it down — is one of the defining challenges of modern engineering.
The High Cost of Decay
Why does corrosion command so much attention? Because its consequences reach far beyond a flaky surface. The impact falls on three critical pillars:
• Safety: This is non-negotiable. A vessel wall that thins unnoticed can rupture without warning, releasing toxic chemicals or flammable gases. Industrial history is littered with catastrophic fires and environmental disasters that trace back to a corroded pipe nobody spotted in time.
• Reliability: Unexpected failures trigger unscheduled shutdowns. For a plant producing thousands of units per hour, even a few hours of downtime can translate into millions in lost revenue — to say nothing of the reputational cost of being seen as unreliable.
• Economics: The financial toll is staggering. Global estimates put the annual cost of corrosion in the trillions of dollars. Beyond replacing damaged parts, engineers must “overdesign” equipment from the start — adding extra wall thickness as a “corrosion allowance” — and sometimes specify expensive exotic alloys. Even a thin layer of rust on a heat exchanger surface acts as insulation, forcing pumps to work harder and burning more energy every single day.
The Science: Nature’s Invisible Battery
Here is the remarkable thing: corrosion is not chaos. It is elegantly predictable chemistry. At its core, a corroding piece of metal behaves exactly like a battery — one that nature assembles spontaneously wherever the conditions are right.
Four components must all be present at the same time for corrosion to proceed. Remove any one of them, and the process stops entirely. This insight is the foundation of all corrosion control:
• The Anode: The region where metal atoms surrender electrons and dissolve into solution. This is where destruction actually occurs.
• The Cathode: The region that accepts those electrons. It doesn’t corrode itself, but it drives the reaction that destroys the anode.
• The Electrolyte: A conductive liquid — seawater, acidic condensate, even humid air — that allows ions to migrate between anode and cathode.
• The Metallic Path: A physical connection through which electrons travel from anode to cathode, completing the circuit.
The Shield: Passivation
If metal is so eager to corrode, why does a stainless steel kitchen sink stay gleaming for decades? The answer is passivation. Certain metals — stainless steel, aluminum, titanium — react almost instantly with atmospheric oxygen to form a microscopically thin, tightly bonded oxide layer on their surface. Think of it as the metal pulling on an invisible raincoat. As long as that coat stays intact, the environment never reaches the metal underneath. Break it — with a scratch, a chemical attack, or the wrong electrochemical conditions — and trouble begins.
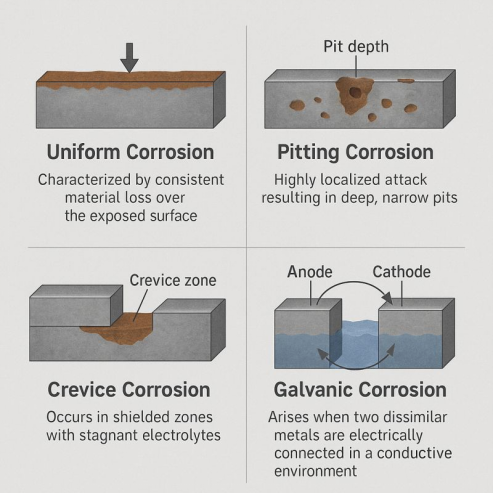
The Rogues’ Gallery: How Corrosion Attacks
Corrosion is a shape-shifter. Knowing which form you are dealing with is half the battle, because each type demands a different detection and prevention strategy.
Uniform Corrosion is the familiar, even thinning of a metal surface — like a sugar cube dissolving in tea. It’s destructive but predictable, and engineers account for it simply by making metal thicker at the design stage.
Pitting and Crevice Corrosion are the most treacherous. Pitting punches deep, narrow holes through a surface that may otherwise look perfectly healthy. A single pit can perforate a pipe wall long before an inspection would flag anything unusual. Crevice corrosion hides in stagnant pockets — beneath gaskets, under bolt heads — where trapped liquid depletes oxygen and creates an intensely corrosive chemical environment, eating away unseen.
Galvanic Corrosion is the price paid for mixing metals. When a “noble” metal like stainless steel sits in electrical contact with an “active” metal like carbon steel in the presence of a liquid, the active metal becomes a willing sacrifice. A brass valve bolted to a steel pipe is a classic trap — the steel around the joint corrodes aggressively while the brass remains pristine.
Stress Corrosion Cracking (SCC) is perhaps the most insidious. It requires a deadly combination: tensile stress in the metal, a susceptible material, and a specific hostile environment. The result is not slow dissolution but sudden, brittle cracking — often with no visible warning. Stainless steel in chloride-rich environments (like coastal seawater) is a notorious victim.
Microbiologically Influenced Corrosion (MIC) reminds us that even biology is in on the act. Certain bacteria and algae colonize metal surfaces and secrete acids or sulfide compounds that create a highly corrosive microenvironment beneath a layer of slime. MIC is a growing concern in water systems, pipelines, and storage tanks where conditions allow biofilms to thrive.
The Achilles’ Heel: Welding
Welding is indispensable — it is how we join the pipes, vessels, and structural members that form our industrial infrastructure. But every weld introduces a vulnerability. The intense heat of welding creates a Heat-Affected Zone (HAZ) in the surrounding metal. In stainless steels, this heat drives chromium — the very element responsible for corrosion resistance — to combine with carbon and precipitate out of the alloy’s structure. The result is a band of metal flanking the weld that is chromium-depleted and far more vulnerable to attack. This process, called sensitization, can turn a weld from a point of strength into a line of weakness.
Winning the War: Prevention and Control
Because corrosion is electrochemistry, the strategy for fighting it is straightforward in principle: break the circuit. In practice, engineers draw on a layered arsenal of defences:
• Intelligent Design: The cheapest fix is the one made on paper. Eliminating crevices, ensuring tanks drain fully, and avoiding joints between dissimilar metals costs nothing at the design stage and prevents enormous problems later.
• Material Selection: Sometimes the environment simply demands a better suit of armour. Upgrading from carbon steel to duplex stainless steel, or from stainless to titanium, may look expensive on a spreadsheet but pay for themselves many times over across a plant’s lifetime.
• Coatings: Paint, epoxy linings, and thermal-spray coatings create a physical barrier that keeps the electrolyte from ever touching the metal. Their weakness is that they must be maintained; a holiday (pinhole) in a coating can actually accelerate localised corrosion underneath.
• Chemical Inhibitors: These are molecules added to the process fluid that adsorb onto the metal surface, reinforcing the passive film or blocking the electrochemical reactions that drive corrosion. They are widely used in oil and gas pipelines and cooling water systems.
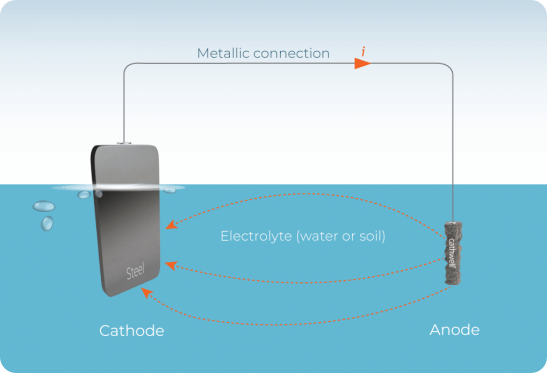
• Cathodic Protection: This is electrochemical judo. By connecting a block of a more active metal — zinc or magnesium — to a structure, engineers force that sacrificial anode to corrode in place of the protected metal. Every ship’s hull, buried pipeline, and offshore platform leg relies on this principle. In impressed current systems, an external power supply takes the role of the sacrificial anode, allowing precise electrical control.
Conclusion
Corrosion is not a problem to be solved; it is a condition to be managed. Thermodynamics guarantees that metals will always seek to return to their lowest energy state, and no coating, alloy, or inhibitor changes that fundamental reality. What engineering does — at its best — is buy time: extending the life of structures by decades, keeping safety margins intact, and ensuring the infrastructure that powers modern civilisation remains trustworthy for the people who depend on it.
In that sense, the battle against corrosion is less like a war to be won and more like a garden to be tended. The moment you stop paying attention, nature quietly takes back what it was always trying to reclaim.

FAQ
Q: Why is corrosion such a big deal in industrial settings?
A: Corrosion threatens three critical areas: safety (corroded pipes can rupture and release hazardous materials), reliability (unexpected failures cause costly shutdowns), and economics (global corrosion costs reach trillions annually). It's not just cosmetic damage—it's an existential threat to infrastructure that requires constant management.
Q: What exactly causes metal to corrode?
A: Corrosion works like a natural battery. Four components must be present: an anode (where metal dissolves), a cathode (which accepts electrons), an electrolyte (conductive liquid like seawater), and a metallic path connecting them. Remove any one element and corrosion stops—this principle guides all prevention strategies.
Q: Can corrosion ever be completely prevented?
A: No. Thermodynamically, refined metals naturally want to return to their ore state. Engineers can't stop corrosion, only slow it down through material selection, coatings, cathodic protection, and smart design. It's about managing the inevitable decay and buying decades of safe, reliable service life.
Keep Learning
Heating Slabs, Blooms, and Rectangular Bars by Induction: Edge Control Is Everything
Key Takeaways Edge control is critical: Slabs, blooms, and rectangular bars exhibit complex electromagnetic edge and end effects that directly impact heating uniformity and product quality. Frequency selection matters: The optimal skin-depth-to-thickness ratio ($d/\delta$) of approximately 3–3.5 for nonmagnetic slabs balances penetration depth, heating uniformity, and coil electrical efficiency. Coil overhang optimization: A normalized coil overhang of about $\sigma/d \approx 0.7$ creates balanced pow......
Induction Heating for Semisolid Processing: Billet and Slug Recipes That Work
Key Takeaways Semisolid Temperature Window: Aluminum alloys require heating to 575°C–595°C with ±3°C to ±4°C uniformity for consistent 50% liquid fraction and high-quality castings. Billet Geometry Matters: Maintain slug length-to-diameter ratios between 1:2 and 1:3 to prevent segregation and ensure uniform heating throughout the workpiece. Multistage Heating Is Essential: Combine rapid power-coil heating with soaking in holding coils to homogenize temperature and microstructure before casting. Freque......
Billet Heating and End Heating by Induction: Uniformity, Throughput, and Coil Strategy
Key Takeaways Temperature Targets: Steel billets require heating to 1050°C–1260°C with surface-to-core uniformity within ±20°C to ±30°C for quality forging outcomes. Frequency Matters: Billet diameter dictates frequency selection—higher frequencies (>30 kHz) for small billets, lower frequencies (<500 Hz) for large billets—to optimize heating depth and uniformity. Profiled Coils Outperform: Variable-pitch, graded coils deliver superior temperature uniformity and shorter coil lengths compared to convent......
In-Line Induction Heating of Bars and Rods: Continuous Throughput Without Temperature Surprises
Key Takeaways Electromagnetic Skin Effect: During initial heating, eddy currents concentrate in a thin surface layer, creating steep surface-to-core temperature gradients that must be managed carefully. Arcing Prevention: Longitudinal voltage drops along bars can cause arcing with support rolls. Alternating coil winding directions or tailored electrical connections are key mitigation strategies. Copper Shield Plates: Unslotted copper end plates act as Faraday rings to reduce electromagnetic cross-talk......
Fundamentals and Design Approaches for Induction Mass Heating
Key Takeaways Frequency is critical: The workpiece diameter-to-penetration depth ratio must exceed four to prevent eddy current cancellation and ensure efficient, uniform heating through the cross section. Material-specific challenges: Stainless steels and superalloys demand tighter control due to high electrical resistivity and low thermal conductivity, making cross-sectional uniformity harder to achieve. Four heating modes: Engineers select from static, progressive multistage, continuous, and oscill......
Advanced Induction Joining and Assembly: From Adhesive Bonding to Motor Rotor Integration
Key Takeaways Induction Bonding: Uses heat transferred from an induction-heated metallic part to cure adhesives, enabling strong joints between dissimilar materials like plastics, ceramics, and metals. Cap Sealing: A non-contact induction process that heats an aluminum foil liner inside bottle caps to create hermetic, tamper-evident seals at high line speeds. Shrink Fitting: Exploits thermal expansion to create high-strength interference fits without fasteners or fillers, and is often reversible for m......